|
m is the magnetic quantum number: m are integers that range from -l to l, including zero.a low shell with a high subshell may be higher in energy than a higher shell with a low subshell.for a given shell, higher subshells have higher energy. 
A generalized formula for the above pattern: for any subshell, 4l+2 electrons can be held.each orbital holds a maximum of 2 electrons.l is the angular momentum quantum number: l are integers that range from 0 to n-1.Quantum numbers l, m, s, and number of quantum states (electrons) per orbital.The emission spectrum shifts to a slightly longer wavelength.The absorption spectrum corresponds to the emission spectrum in pattern.The emission spectrum looks like colored lines on a black background.The emission spectrum shows what wavelengths of light are emitted.The absorption spectrum looks like black lines on a rainbow background.The absorption spectrum shows what wavelengths of light are absorbed.Excited state electrons come back down to the ground state via release of energy.Excited states are higher in energy than ground states.When they absorb energy, they get promoted to excited states.Electrons are normally in their ground state.Thus, there are 2n^2 electrons per shell.There are n squared orbitals per shell.Higher n shells are higher in energy (if subshells are the same).The principle quantum number, n, defines what shell the electron is in.In quantum mechanics, hydrogen electron exists in a spherical probability cloud around the nucleus.In the Bohr model, the hydrogen electron orbits the nucleus.Orbital structure of hydrogen atom, principal quantum number n, number of electrons per orbital.Clipart and elements found in this PDF are copyrighted and cannot be extracted and used outside of this file without permission or license.įiles are partially or fully non-editable to protect the images that are copyrighted and purchased through licenses. If you and a team of teachers would like to use this resource together, please purchase additional licenses on the resource purchase page.įailure to comply with these terms of use is a copyright infringement and a violation of the Digital Millennium Copyright Act (DMCA). You are not allowed to distribute this digital resource to other teachers or post this resource on any webpage or server that is available for public view. This resource is for you, the purchaser, alone. * place this file on your own password-protected class page or server (Blackboard, Google Drive, etc) AS LONG AS no other teacher has access to that class webpage. Purchasing my teaching resources allows you to: If you have any questions or concerns, please reach out to us on the question and answer section of my store and we will get back to you quickly! 
The PDF file contains the student page, the answer key, and my terms of use page.Ĭheck out my Chemistry Doodle Notes for some engaging resources that thoroughly explain Chemistry concepts in a versatile way! 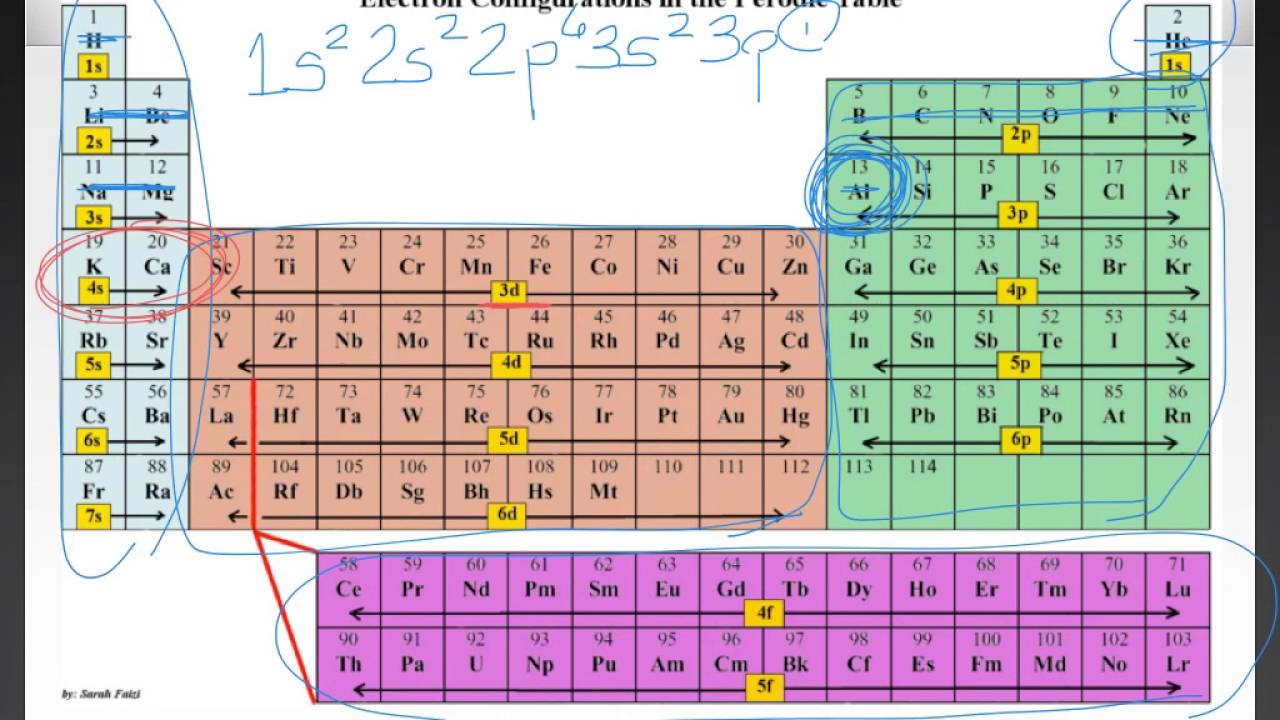 
This page is also part of a Periodic Table Trends Homework Unit. Click here to read about that whole year bundle if you want to buy a whole year’s worth of homework pages at a discount as compared to buying them separately. This page is part of my giant Chemistry Homework for a Year Bundle. This is great for basic, regular, and higher level learners. The questions ask students to identify which block an element belongs to. This chemistry homework page is perfect for students to examine the electron configuration blocks on the periodic table.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
